- ABOUT JIRA
-
- Chairman Message
- Business and About JIRA
- Business & Main Activities
- Product of Scope
- History
- Organization
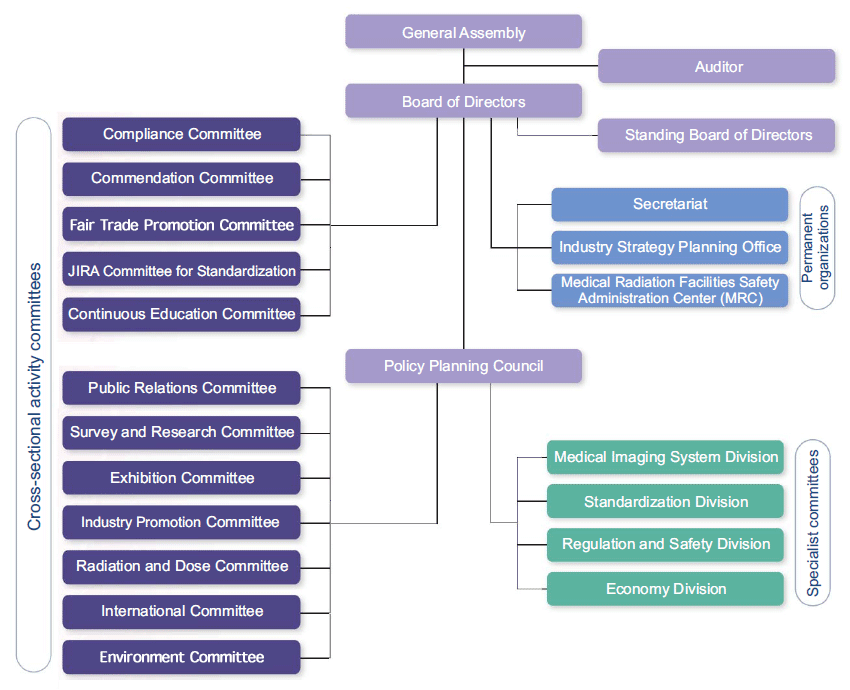
- Organization Chart
- Officers
- Member Companies
- Business Activities
- JIRA Vision 2030
- Activity in 2024
- Compliance
- Code of Ethics
- Contact Us
- Access to JIRA
- DIVISION & COMMITTEE
-
- Divisions
- Medical Imaging System Division
- Standardization Division
- Regulation and Safety Division
- Economy Division
- Permanent Organization
(Reporting to BoD) - Secretariat
- Industry Strategy Planning Office
- Medical Radiation Facilities Safety Administration Center
- Committees
- Public Relations Committee
- Survey and Research Committee
- Exhibition Committee
- Industry Promotion Committee
- Radiation and Dose Committee
- International Committee
- Environment Committee
- Compliance Committee
- Fair Trade Promotion Committee
- Continuous Education Committee
- EXHIBITION
-
- ITEM in JRC(International Technical Exhibition of Medical Imaging)
- Guide to Exhibition
- Information to Exhibitor
- Past Exhibitions
- Annual Scientific Meeting of the Japanese Society of Nuclear Medicine & Exhibition
- Guide to Exhibition
- Information to Exhibitor
- Past Exhibitions
- TOPICS
- Old Site